Across the life sciences industry, stability programs are essential for demonstrating the safety, quality, and shelf life of pharmaceutical and medical products. These studies generate critical data that support regulatory submissions and product lifecycle management.
The FDA has consistently developed regulatory guidance to encourage the industry to utilize digitalized systems. Nevertheless, many organizations continue to manage stability studies through paper documentation, spreadsheets, and disconnected tools that make long-term study management difficult and increase regulatory risk.
Transitioning to a digital stability management platform can significantly improve efficiency, data integrity, and regulatory compliance. However, successful implementation requires careful planning on the client side before engaging with a software vendor. Organizations that take the time to build the right internal project structure and clearly define their requirements are far more likely to select and implement a system that meets their long-term needs.
A successful project begins with the formation of a cross-functional internal project team responsible for establishing the vision for the system and guiding the vendor selection and implementation process.
Establishing an Internal Project Team
Before evaluating potential software solutions, organizations should establish a dedicated project team responsible for defining the goals and requirements of the new system. This team should represent all groups that will interact with the system or support it during its lifecycle.
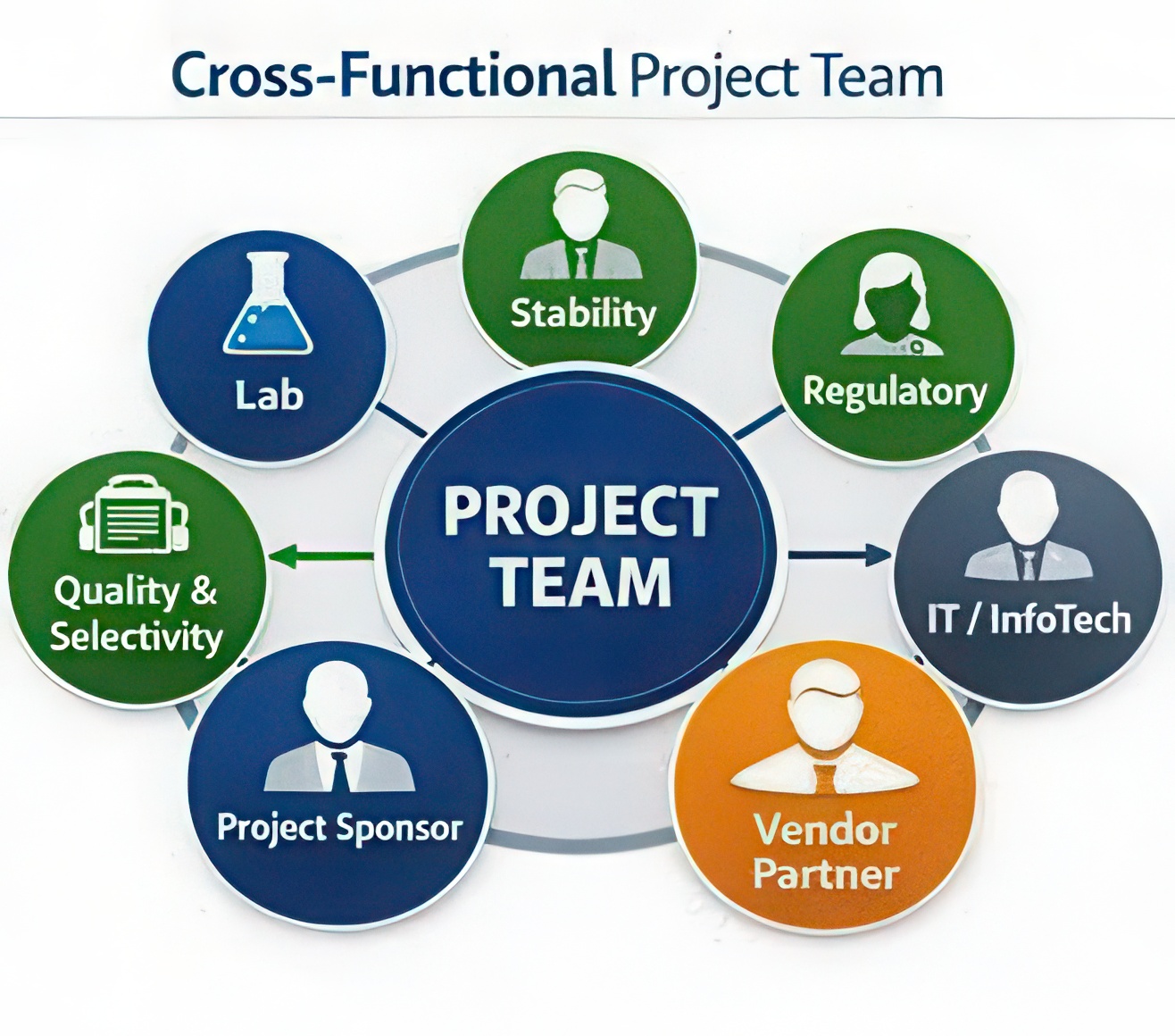
A stability management system touches multiple functions across an organization, including laboratory operations, regulatory compliance, data analysis, and IT infrastructure.
As a result, the project team should include representatives from each of these areas to ensure that the system meets operational, technical, and regulatory expectations.
Typically, the project team includes members from:
Stability or analytical laboratory teams
These users represent the primary operators of the system. They understand the daily workflows involved in managing stability protocols, sample pulls, testing schedules, and data entry. Their role is to ensure the system supports real laboratory processes.
Because stability studies rely heavily on statistical evaluation to determine product shelf life, organizations benefit from including individuals responsible for stability data analysis. These team members can help evaluate the statistical tools available within potential software solutions and determine whether the system supports regulatory expectations such as trend analysis, regression modeling, and shelf-life prediction.
Quality assurance and regulatory representatives
Quality and regulatory participants play a critical role in ensuring that the selected system supports the organization’s broader compliance obligations. Their responsibility is to evaluate whether the platform aligns with applicable regulatory expectations, including data integrity principles, electronic record requirements, and global regulatory frameworks such as FDA regulations, EU GMP requirements, and ICH stability guidelines (including the ICH Q1 series).
These team members help ensure that the system supports appropriate audit trails, electronic signatures, controlled workflows, and documentation management required for regulated environments. They also define system validation expectations and ensure that the platform can support regulatory inspections, stability reporting, and long-term data retention requirements.
By participating early in the selection process, quality and regulatory representatives help ensure that the chosen solution not only meets operational needs but also supports the organization’s compliance strategy across global regulatory authorities.
Information technology (IT)
IT representatives play an important role in evaluating the technical aspects of a proposed system. Their responsibilities typically include assessing system architecture, infrastructure requirements, security controls, and compatibility with the organization’s existing technology environment. IT teams also review integration capabilities to determine how the system may connect with other enterprise platforms such as document management systems, enterprise resource planning (ERP) tools, or laboratory instrumentation.
While IT involvement is essential, it is equally important that system selection is not driven by IT alone. Stability management systems must support complex scientific workflows, regulatory requirements, and long-term study management processes that are best understood by laboratory and quality professionals. If IT alone selects the platform based purely on technical criteria, there is a risk that the system may not adequately support real laboratory operations or stability program requirements.
For this reason, IT should function as a technical advisor within the broader cross-functional project team, ensuring the system meets infrastructure and security standards while laboratory, quality, and regulatory stakeholders evaluate whether the platform supports operational workflows, statistical analysis, and regulatory compliance needs. A balanced evaluation approach ensures that the selected system is both technically sound and operationally effective for the teams who will use it daily.
Project leadership or program management
A project leader or sponsor helps coordinate activities, maintain timelines, and ensure the project aligns with organizational priorities. This role is often filled by a senior laboratory manager or R&D leader.
By assembling a team with diverse expertise, organizations ensure that the system selection process considers all operational and regulatory perspectives.
Defining the Vision and Requirements
Once the project team is established, the next step is to clearly define the goals and requirements for the new system. This phase is critical because it sets the foundation for vendor evaluation and system selection.
The team should begin by mapping current stability workflows. This includes documenting how protocols are created, how samples are stored and tracked, how pull schedules are managed, and how stability data is analyzed and reported. Identifying inefficiencies in current processes helps clarify what the new system must improve.
From this analysis, the team can begin developing user requirements that describe how the system should function. These requirements are often categorized into three levels:
Mandatory requirements describe capabilities that the system must provide in order to meet regulatory or operational needs.
Desirable requirements describe features that improve efficiency or usability but are not essential.
Optional requirements include additional capabilities that may provide value but are not critical for implementation.

For stability programs, typical requirements may include:
The final output of this phase should be a documented set of user requirements and a clear project charter that defines the objectives, scope, and expected outcomes of the project.
Evaluating Stability-Specific Software Solutions
Once the organization has defined its requirements, the project team can begin evaluating potential software vendors. Many organizations use a structured Request for Proposal (RFP) process to compare solutions.
During vendor evaluations, it is important for the team to understand the difference between a generic laboratory information management system (LIMS) and a dedicated stability management platform.
Generic LIMS platforms are designed to manage routine laboratory operations such as sample logging and test result reporting. While they can be adapted for stability studies, they often require significant customization and may lack specialized functionality. Additionally, since the generic LIMS do not have the inherent statistical analysis tool, the data must be exported to third party statistical tool that substantially increases the risk to data integrity as there will be no audit trails for the activities performed outside the LIMS.
Dedicated stability management platforms, such as Novatek Stability-LIMS, are designed specifically to manage the complex workflows associated with long-term stability studies. These systems include functionality for automated pull calendars, sample tracking, protocol management, and integrated statistical analysis that considers several sample time points and lots for the product shelf life determination.
When evaluating vendors, the project team should focus on how well each solution supports the organization’s defined requirements. Product demonstrations provide an opportunity to see how stability protocols are created within the system, how sample inventory is tracked, and how stability data is analyzed.
Evaluating Built-In Statistical and Shelf-Life Prediction Tools
One of the most important capabilities of a stability management system is its ability to analyze stability data and predict product shelf life. Stability studies generate large volumes of data over extended periods, and regulatory authorities expect organizations to apply scientifically sound statistical methods when determining product expiration dating.
For this reason, organizations should carefully evaluate the statistical tools built into potential software solutions.
Advanced stability management platforms provide integrated tools for regression analysis, trend evaluation, and shelf-life prediction. These tools allow scientists to analyze stability data directly within the system rather than exporting data to spreadsheets or external statistical software.
Systems may also support accelerated aging analysis using Arrhenius modeling, allowing teams to estimate product shelf life based on data generated under accelerated storage conditions. These predictive capabilities help organizations make earlier and more informed decisions about product shelf life and regulatory strategy.
By incorporating statistical tools directly into the stability platform, organizations can reduce manual data handling, improve traceability, and ensure that analytical methods align with regulatory expectations.
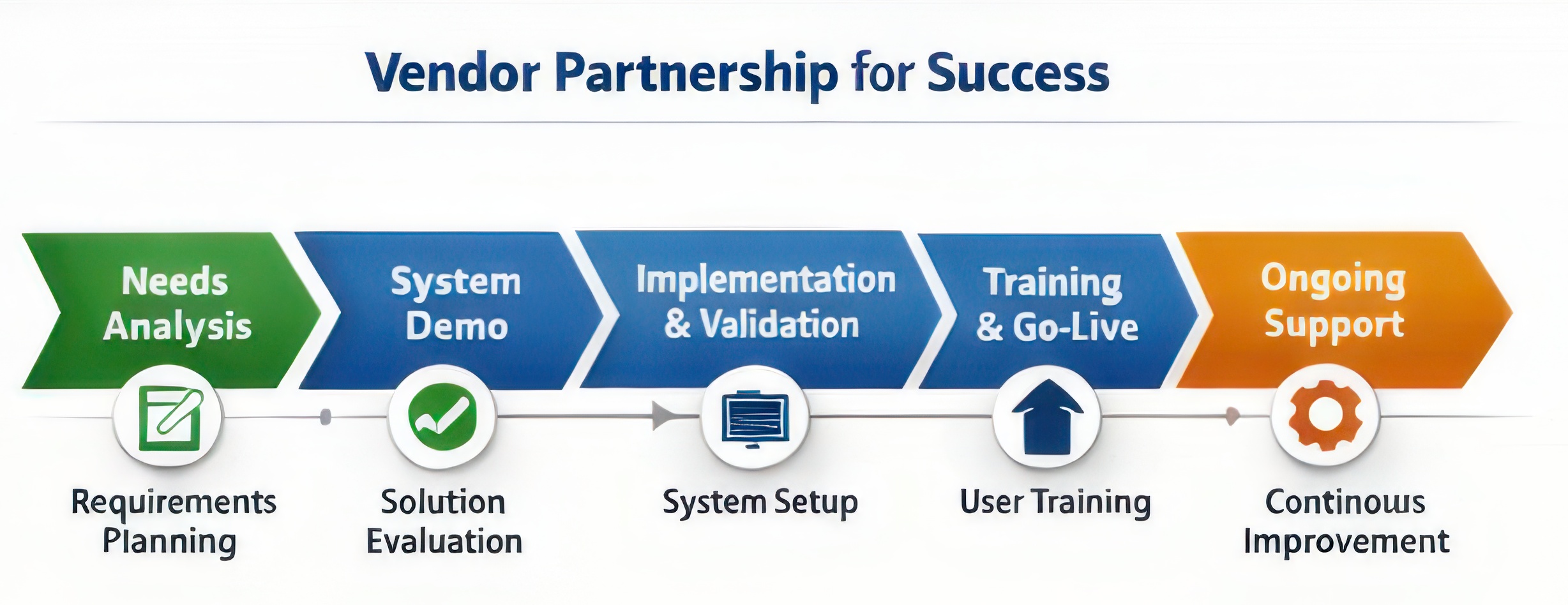
Partnering with the Vendor During Implementation
Once a vendor has been selected, the organization and vendor work together to implement the system. This phase requires continued collaboration between the internal project team and the vendor’s implementation specialists.
Because stability management systems are used in regulated environments, implementation includes a structured system validation process. This typically includes Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ).
During this phase, system workflows are configured to match the organization’s stability processes. Standard operating procedures are updated to reflect the new digital workflows, and strategies for handling legacy stability data are developed.
Training is another critical component of implementation. The vendor typically provides structured training sessions to ensure laboratory staff understand how to operate the system and incorporate it into their daily workflows.
Maintaining a Long-Term Vendor Partnership
Successful digital transformation does not end once the system goes live. Organizations benefit most when they maintain an ongoing partnership with their software vendor.
As per FDA requirements, vendors must continue to provide technical support, maintenance and system updates, and guidance on optimizing system functionality and ensuring the system meets the data integrity and regulatory compliance at all times. This partnership becomes particularly valuable as regulatory expectations evolve further.
Recent revisions and ongoing updates to ICH stability guidelines, including changes to the broader ICH Q1 series, highlight increasing expectations around stability data trending and statistical evaluation. Vendors play an important role in ensuring their platforms evolve alongside these regulatory developments.
By maintaining systems with robust statistical tools, advanced trending capabilities, and flexible reporting options, stability management platforms can help organizations remain aligned with evolving global regulatory standards.
Benefits of Moving Stability Programs into the Digital Era
The transition from paper-based stability management to a fully digital system represents far more than a simple technology upgrade. It is a fundamental shift in how organizations manage stability studies, interpret scientific data, and maintain regulatory compliance throughout the product lifecycle. As stability programs grow in complexity and regulatory expectations continue to evolve, manual processes built around spreadsheets and paper documentation increasingly limit efficiency, transparency, and data integrity.
Digital stability management platforms provide organizations with the tools needed to centralize stability workflows, automate routine tasks, and analyze stability data in real time. Features such as automated pull schedules, electronic protocol management, integrated statistical analysis, and built-in shelf-life prediction tools allow stability teams to focus more on scientific interpretation and less on administrative tasks. At the same time, digital systems strengthen compliance by providing structured workflows, audit trails, and controlled data environments aligned with global regulatory expectations.
However, the success of a digital transformation effort depends heavily on the preparation and collaboration that occur before and during system implementation. Organizations that assemble strong internal project teams, clearly define their operational and regulatory requirements, and engage collaboratively with experienced vendors are best positioned to select a solution that supports both their current needs and their long-term growth.

Ultimately, digitalization enables stability programs to operate with greater efficiency, scientific rigor, and regulatory confidence. By replacing fragmented manual workflows with integrated digital platforms, organizations gain improved visibility into stability data, stronger control over study management, and the ability to respond more effectively to evolving regulatory expectations.
In an increasingly data-driven regulatory environment, modern digital stability management systems are becoming an essential foundation for maintaining compliant, efficient, and future-ready stability programs.
Share This Article with the Stability Community!
May 2, 2026
From conception to destruction, there are numerous CGMPs governing the life cycle of a sample, with multiple professionals, departments and organizations having a hand in [...]
February 28, 2026
Ben Was Right QC and analytical labs are facing heightened expectations as method lifecycle principles, data integrity, and digital lab tools reshape the regulatory [...]
February 6, 2026
If you oversee or participate in a medical product Stability program, you have been, or will be involved in an audit or inspection. While inspections [...]
Share your questions and experiences
A stabilitarian encounters new situations every day. StabilityHub’s discussion forums give Stabilitarians an opportunity to ask questions and offer solutions to specific scenarios. Join in the conversations with other Stabilitiarians and share your knowledge!
A stabilitarian encounters new situations every day. StabilityHub’s discussion forums give Stabilitarians an opportunity to ask questions and offer solutions to specific scenarios. Join in the conversations with other Stabilitiarians and share your knowledge!





