Hollister Incorporated, a global, employee-owned healthcare manufacturer, began a critical modernization initiative to overhaul its stability program. For years, Hollister had relied on traditional, paper-based systems to manage complex stability studies supporting the development and regulatory approval of its medical products. These methods, while long-standing, had become increasingly burdensome as regulatory expectations, product portfolios, and data volumes grew. To address these challenges, Hollister embarked on a structured digital transformation—moving from a fragmented paper workflow to a fully digital, dedicated Stability Management System.
A Paper-Based Past
Hollister’s legacy stability system was built around manual processes that had changed little over time. Stability protocols were drafted and routed in hard copy for approval. Analysts created and maintained Excel spreadsheets for test results and pull calendars, executed tests, and manually entered data for each time point. Statistical analysis often required weeks of effort, and final reports were printed, signed, scanned, and stored in filing cabinets. Information was dispersed across multiple locations, frequently duplicated or inconsistent.
While these processes had supported the company for years, they also created significant operational and compliance challenges. The lack of an integrated audit trail raised concerns about data integrity and made it difficult to meet regulatory requirements. Manual workflows were time-consuming and prone to transcription errors, while real-time visibility into study status was limited. Tracking samples and scheduling pulls required constant manual oversight, and reporting remained static and reactive. As regulatory frameworks such as 21 CFR Part 11 and data integrity requirements increasingly emphasized data traceability, electronic records, and system validation, the paper-based approach no longer met Hollister’s compliance and efficiency needs.
A Strategic Decision to Go Digital
Hollister recognized that incremental improvements to its existing workflows would not be sufficient. Rather than adopting a generic laboratory information management system, the company chose to implement a dedicated stability management platform—a solution purpose-built to address the unique operational and regulatory demands of stability programs. Unlike general laboratory workflows, stability studies involve multi-year timelines, complex testing schedules, inventory control, and rigorous regulatory reporting requirements. A one-size-fits-all LIMS could not deliver the level of specialization and control Hollister needed.
The transformation began with a comprehensive process audit led by an external consultant. The audit confirmed what Hollister’s teams already understood: the legacy system was inefficient, non-validated, and not aligned with evolving regulatory standards. The consultant recommended implementing a validated stability management platform that can manage protocols, automate pull schedules, provide built-in trending and statistical analysis, and maintain a full audit trail.
Stakeholder Engagement
After Executive sponsorship secured funding and set clear priorities, a cross-functional team—drawing from Stability, IT, R&D, and Quality—was formed to foster collaboration, share expertise, and ensure every stakeholder’s needs were addressed. This approach empowered stakeholders with greater transparency, influence over project outcomes, and solutions tailored to their unique requirements. Building on this foundation of strong sponsorship and collaborative teamwork, the project moved into the next critical phase: system selection and requirements gathering.
Why a Dedicated Stability Platform Matters
One of Hollister’s most critical strategic choices was to select a stability-specific system rather than a general laboratory system. Generic LIMS platforms typically focus on broad laboratory activities such as sample logging, result entry, and reporting, but lack functionality tailored to long-term stability testing.
A dedicated stability management system offers specialized capabilities that directly support stability workflows. These include automated pull calendar generation with configurable alerts, integrated barcode labeling and inventory control for precise sample tracking, and stability protocol templates that ensure standardized, compliant study design. Built-in statistical analysis and trending tools—such as regression modeling, shelf-life projections, and accelerated aging calculations—eliminate the need for external spreadsheets and manual calculations. Additionally, comprehensive audit trails, electronic signatures, and version control align the system with 21 CFR Part 11 / EU Annex 11 requirements and data integrity regulations. These features allow stability teams to operate more efficiently, with greater accuracy and regulatory confidence than would be possible with a generic laboratory platform. Recognizing these benefits, Hollister’s cross-functional project team identified these capabilities as mandatory user requirements during system selection. By doing so, the team ensured that the chosen system would not only address current operational challenges but also support long-term compliance and scalability.
Selecting a Stability-LIMS
Following a detailed requirements-gathering process and a competitive Requisition for Proposal (RFP), Hollister selected a Stability-LIMS System. The chosen vendor’s system distinguished itself by offering a mature, stability-focused solution with a proven track record in regulated industries. Unlike generic systems that required extensive customization, the selected solution came with pre-configured functionality purpose-built for stability workflows.
The platform provided Hollister with advanced capabilities, including automated protocol management, pull calendar generation, real-time sample tracking, integrated barcode labeling, and sophisticated trending and reporting. It also featured robust audit trails, electronic signature support, and compliance controls aligned with FDA and EU regulatory expectations. The selected system’s seamless integration with Hollister’s existing IT infrastructure further strengthened its position as the ideal solution for the company’s modernization initiative. Figure 1 below provides a visual overview of Hollister’s digital transformation and key system features of the Stability LIMS.
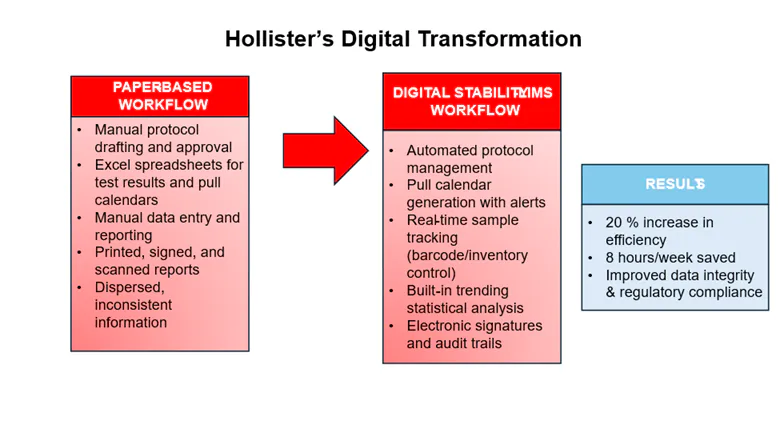
Figure 1
Implementation and Validation
Once selected, the Stability-LIMS underwent a rigorous validation process including Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ). This ensured the platform met both operational requirements and global regulatory standards. At the same time, Hollister developed new standard operating procedures, business continuity and disaster recovery plans, and a clear strategy for managing legacy data. Comprehensive training sessions were held for all stakeholders to ensure smooth adoption of the new system.
The go-live process proceeded smoothly, reflecting careful planning and strong cross-functional collaboration. Paper-heavy workflows were replaced with automated digital processes. Tasks such as pull scheduling, which had previously required manual spreadsheet management, were now automated. Data entry, trending, and reporting were performed directly in the system, with full auditability and real-time visibility. Protocol approvals moved from paper signatures to controlled electronic workflows.
Real-World Impact
The benefits of the transition became immediately evident. For Hollister’s VaProTM Hydrophilic Intermittent Catheter—a flagship product used by patients with spinal cord injuries and other urological conditions—the stability program became significantly more efficient. Multiple testing parameters, including lubricity, bond strength, and packaging integrity, were managed more effectively. Built-in analytical tools enabled the team to apply accelerated-aging models, such as the Arrhenius equation, directly within the platform, streamlining shelf-life estimation and strengthening regulatory submissions.
By moving to the Stability LIMS System, Hollister achieved a 20 percent increase in operational efficiency, saving approximately eight hours of manual work each week. More importantly, the company gained stronger data integrity, improved regulatory compliance, centralized oversight, and the ability to make proactive, data-driven decisions.
Lessons for the Industry
Hollister’s successful implementation of its Stability-LIMS highlights several key lessons for other organizations. Stability testing presents unique operational and compliance challenges that are best addressed with specialized software rather than generic laboratory systems. Defining precise user requirements and aligning cross-functionally are essential for selecting the right platform. Thorough validation and structured deployment support both compliance and operational excellence. And perhaps most importantly, moving from paper to digital is not just a technology shift—it is a strategic transformation that modernizes how stability programs operate.
By embracing its Stability-LIMS, Hollister replaced its fragmented, paper-based processes with a modern, fully digital platform. This transformation has positioned its stability program to meet today’s rigorous regulatory expectations while providing the flexibility and efficiency needed to support continued innovation and growth well into the future.
References
Pack, L. (2024, June 4–6). Essential statistics for stabilitarians: Research questions for stability [Conference session]. KENX Laboratory University – Stability Testing & Program Management, Philadelphia, PA, United States.
Cleary, S., Famili, P., Jorge, P., Routh, W., & O’Neill, J. (2024, September 7). Digitized vs digitalization – What is the difference? StabilityHub. https://stabilityhub.com/2024/09/07/digitized-vs-digitalization-what-is-the-difference/
Cleary, S., Latoz, C., & O’Neill, J. (2025, January 4). Mitigating the risks – Digitalization of stability sample management. StabilityHub. https://stabilityhub.com/2025/01/04/mitigating-the-risks-digitalization-of-stability-sample-management/
Cleary, S., & O’Neill, J. (2025, March 30). Optimize your stability statistics program for digitalization success. StabilityHub. https://stabilityhub.com/2025/03/30/optimize-your-stability-statistics-program-for-digitalization-success/
Novatek International. (n.d.). NOVA-Stability – Stability management software. https://ntint.com/product/nova-stability/
O’Neill, J. (2024, February 5). Can you count on your stability sample inventory? Building a reliable system. StabilityHub. https://stabilityhub.com/2024/02/05/can-you-count-on-your-stability-sample-inventory-building-a-reliable-system/
Share This Article with the Stability Community!
March 28, 2026
Across the life sciences industry, stability programs are essential for demonstrating the safety, quality, and shelf life of pharmaceutical and medical products. These studies [...]
February 28, 2026
Ben Was Right QC and analytical labs are facing heightened expectations as method lifecycle principles, data integrity, and digital lab tools reshape the regulatory [...]
February 6, 2026
If you oversee or participate in a medical product Stability program, you have been, or will be involved in an audit or inspection. While inspections [...]
Share your questions and experiences
A stabilitarian encounters new situations every day. StabilityHub’s discussion forums give Stabilitarians an opportunity to ask questions and offer solutions to specific scenarios. Join in the conversations with other Stabilitiarians and share your knowledge!
A stabilitarian encounters new situations every day. StabilityHub’s discussion forums give Stabilitarians an opportunity to ask questions and offer solutions to specific scenarios. Join in the conversations with other Stabilitiarians and share your knowledge!





