Every stability study has one (unless maybe you are a stability study featured in a Warning Letter). A protocol captures the design of a stability study, but should it 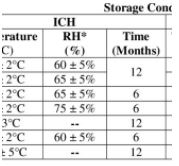
When searching for a document that best describes the design of your stability study, what better place to go than a well-constructed protocol; built on science, compliance, statistics, timing and nature of registration and justification?
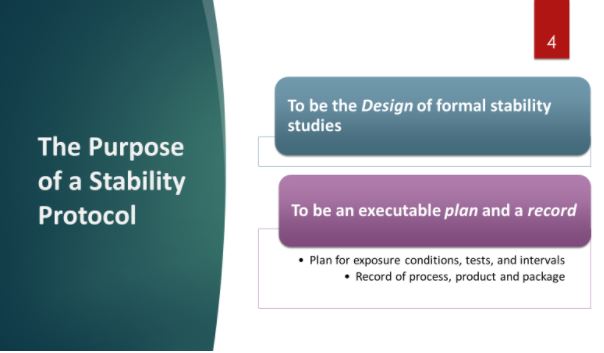
On the other hand, if we fail to build a solid protocol and not manage change control, we risk:
- Disconnects to the “story” of our product
- Ambiguity of process and container-closure details
- Limited scope of design, omitting special international requirements
- Missing input from supporting key disciplines
- Missing revisions and changes in aspects such as Test Methods and Specifications
- Late additions of Contract Research Organizations may not be noted and tracked.
- Receiving a regulatory citation or Warning Letter that announces our protocol failures to the world
As stabilitarians, what elements of quality can we add to our own corporate stability protocols? Chime in (with a free account) at our discussion board!
John O’Neill
Editor, StabilityHub
Share This Article with the Stability Community!
May 2, 2026
From conception to destruction, there are numerous CGMPs governing the life cycle of a sample, with multiple professionals, departments and organizations having a hand in [...]
February 28, 2026
Ben Was Right QC and analytical labs are facing heightened expectations as method lifecycle principles, data integrity, and digital lab tools reshape the regulatory [...]
November 28, 2025
Hollister Incorporated, a global, employee-owned healthcare manufacturer, began a critical modernization initiative to overhaul its stability program. For years, Hollister had relied on traditional, [...]
Share your questions and experiences
A stabilitarian encounters new situations every day. StabilityHub’s discussion forums give Stabilitarians an opportunity to ask questions and offer solutions to specific scenarios. Join in the conversations with other Stabilitiarians and share your knowledge!
A stabilitarian encounters new situations every day. StabilityHub’s discussion forums give Stabilitarians an opportunity to ask questions and offer solutions to specific scenarios. Join in the conversations with other Stabilitiarians and share your knowledge!





